|
10/13/2023 0 Comments Potassium reactivity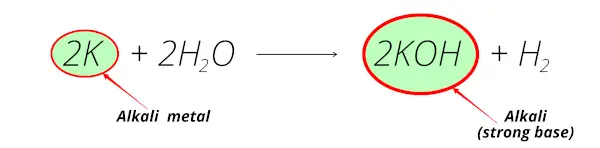
The force of attraction between the nucleus and the electron is less than for smaller atoms, and the outer electron is lost more easily. Potassium can form unstable and highly reactive peroxides if stored for extended periods of time. Water and conventional ABC fire extinguishers can intensify a fire involving potassium and should never be used. It is highly corrosive to eyes, skin and mucous membranes. The larger the atom, the further the outer electron is from the nucleus. Potassium can ignite in moist air or because of friction or static sparks. These atoms are larger because they have more shells. Group 1 elements become more reactive as we move down the group. This can be represented by a half equation (higher tier): Lithium, sodium, and potassium all react with water, for example. A sodium atom loses one electron to form a sodium ion table are the metals that are the most active in the sense of being the most reactive. Group 1 metals all react in the same way, because each of them loses an electron to form a positive ion with a stable electronic configuration – a full outer shell.įor example, sodium loses one electron to form a sodium ion, Na +, with a full outer shell. potassium will do this as well but it is hard to see as the reaction is so fast and also potassium burns with a lilac flame and crackles as it explodes at the end of the reaction.sodium also melts to form a silvery-grey ball on the surface of the water.the solution remaining at the end will be colourless.Potassium is more reactive than Carbon on the reactivity series so it. they will all release heat as they react as the reactions are exothermic and they will eventually disappear. Potassium is a more reactive alkali metal than Sodium but less reactive than Rubidium.the metals move about the surface of the water and fizz as hydrogen gas is produced.lithium, sodium and potassium are less dense than water so they float on the surface of the water.There are some common observations and some differences: 
Sodium + water → sodium hydroxide + hydrogenĪll Group 1 metal reacts with water. Violent explosions also have been observed when mixtures of potassium and halogen acids are subject to shock. This is the equation for the reaction between sodium and water: Potassium is highly reactive with halogens and detonates when it contacts liquid bromine. They must be stored under oil to prevent them reacting with the air or water vapour.Īll Group 1 elements react with water to produce a metal hydroxide and hydrogen.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
